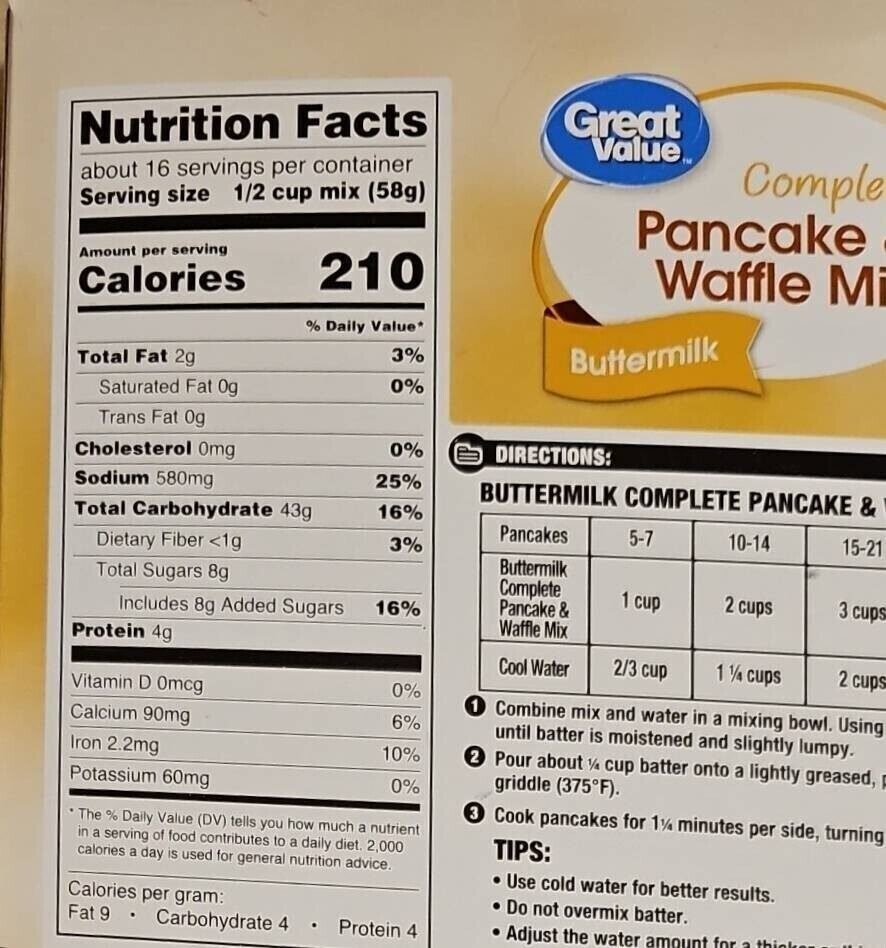
Complete pancake & waffle mix - Great Value - 32 oz
Aquesta pàgina del producte no està completa. Podeu ajudar a completar-la editant-la i afegint-hi més dades a partir de les fotos ja disponibles, o fent-ne més amb l'aplicació de androide o iPhone / iPad. Gràcies!
×
Codi de barres: 0078742370828 (EAN / EAN-13) 078742370828 (UPC / UPC-A)
Quantitat: 32 oz
Marques: Great Value
Propietari de la marca: Wal-Mart Stores, Inc.
Categories: Snacks, Aperitius dolços, Galetes i pastissos, Pastís, Ajudants de cuina, en:Dessert mixes, en:Pastry helpers, en:Baking Mixes, en:Cake mixes, en:Pancake mixes
Botigues: Walmart
Països on es va vendre: Espanya, Estats Units d'Amèrica
Matching with your preferences
Entorn
Empaquetament
Transport
Report a problem
Fonts de dades
Producte afegit per usda-ndb-import
Última modificació de la pàgina del producte per roboto-app.
La pàgina del producte, també editada per acuario, clockwerx, inf, kiliweb, org-app-elcoco, org-database-usda, teolemon, yuka.sY2b0xO6T85zoF3NwEKvlk9dTfT78yLeZxfkp3ak4cqWK77PUM106IffOKs.